Contract packaging service for pharmaceutical products
Our customers produce high-quality pharmaceutical products, such as medications that are used in ophthalmology. Protecting these products with appropriate packaging that meets the regulatory requirements is our duty.
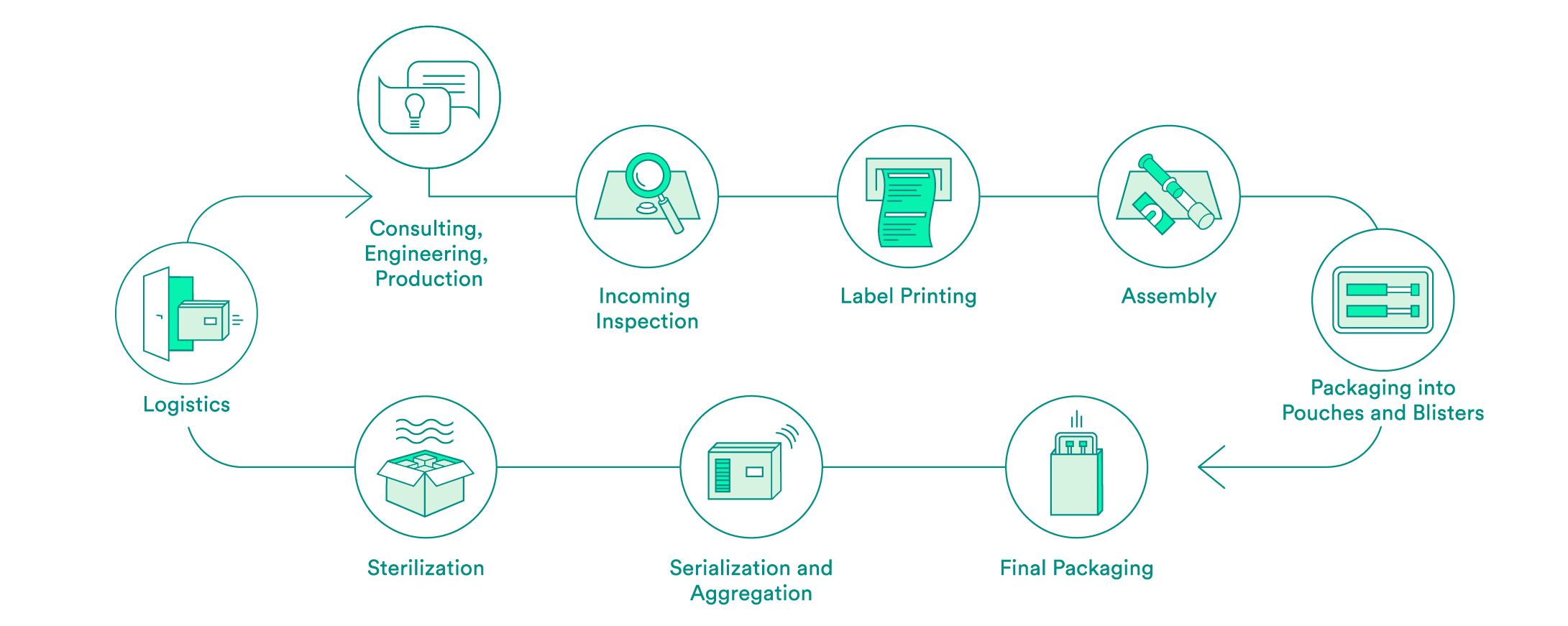
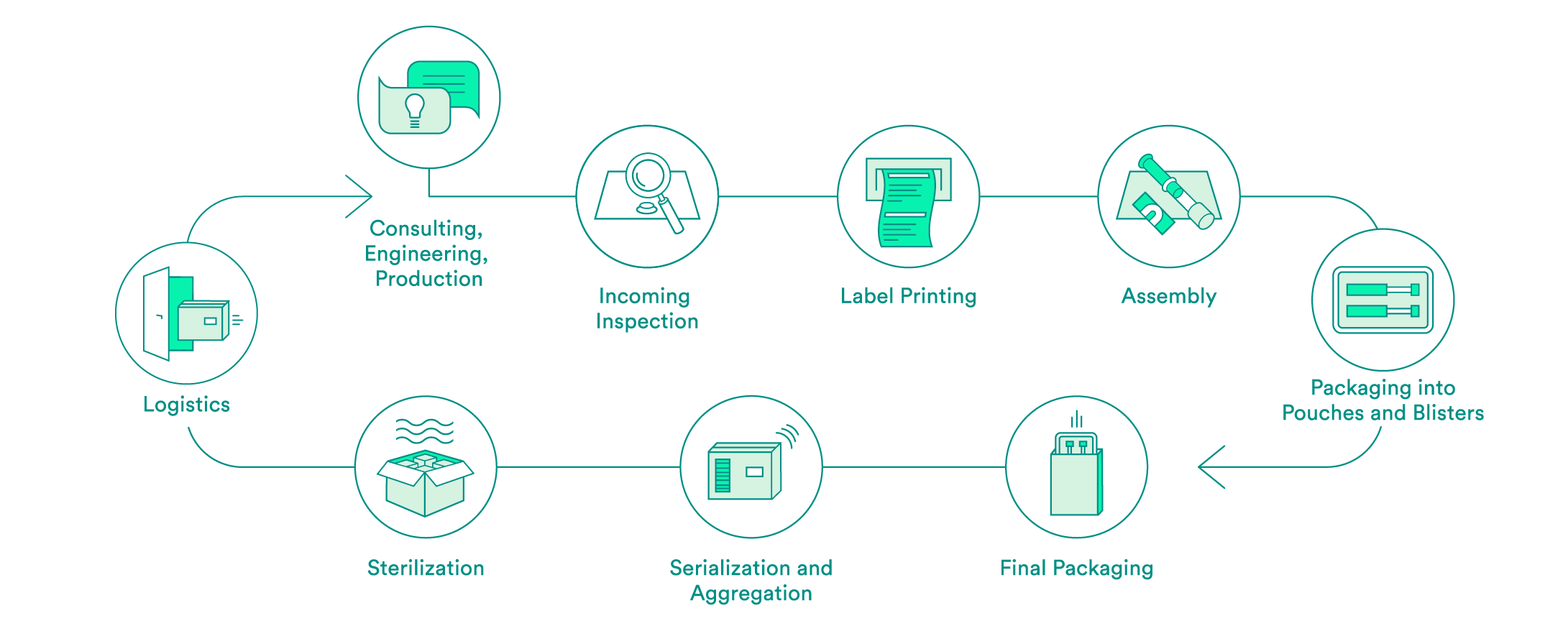
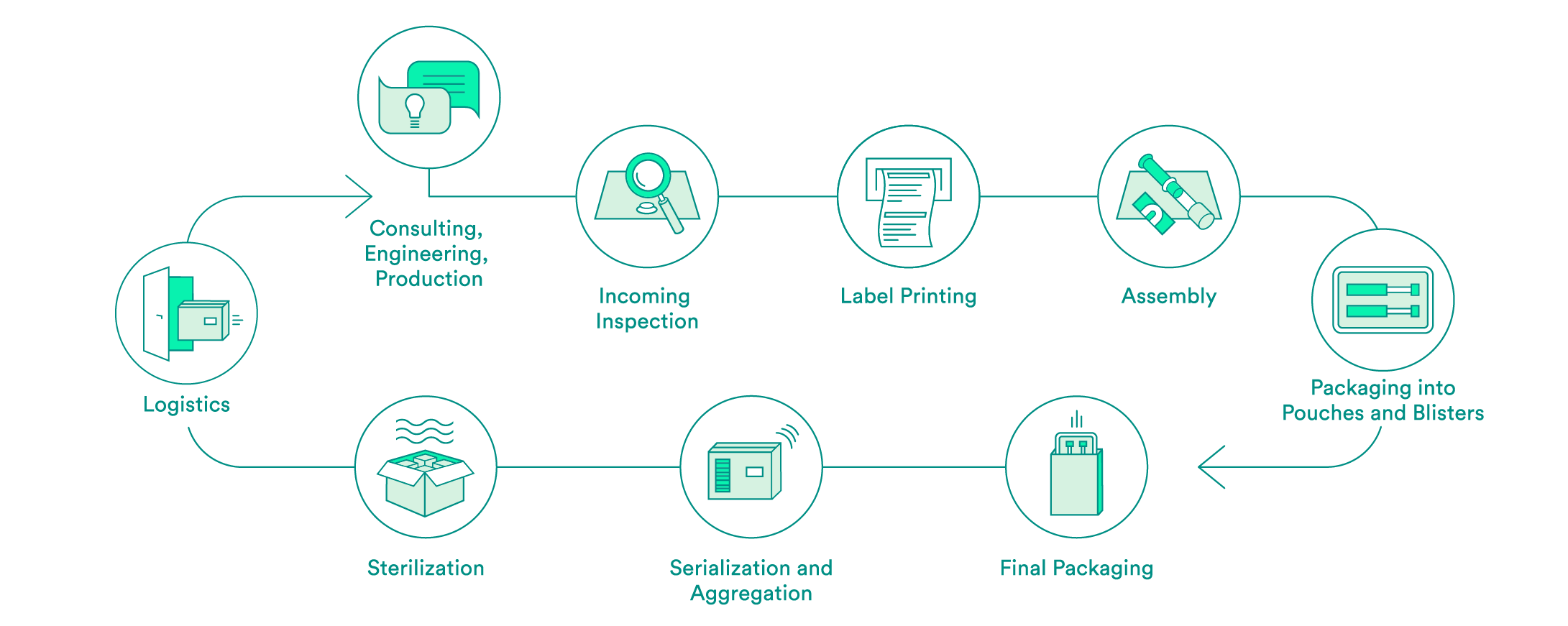
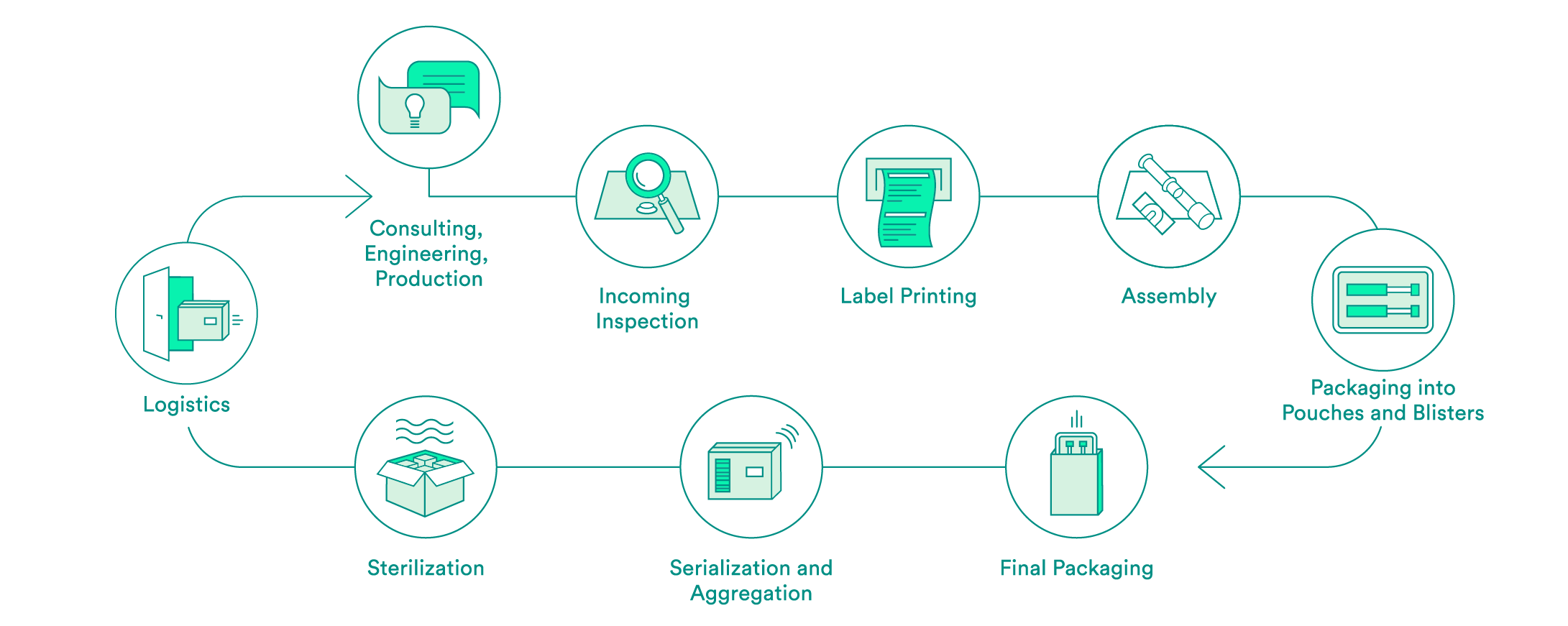
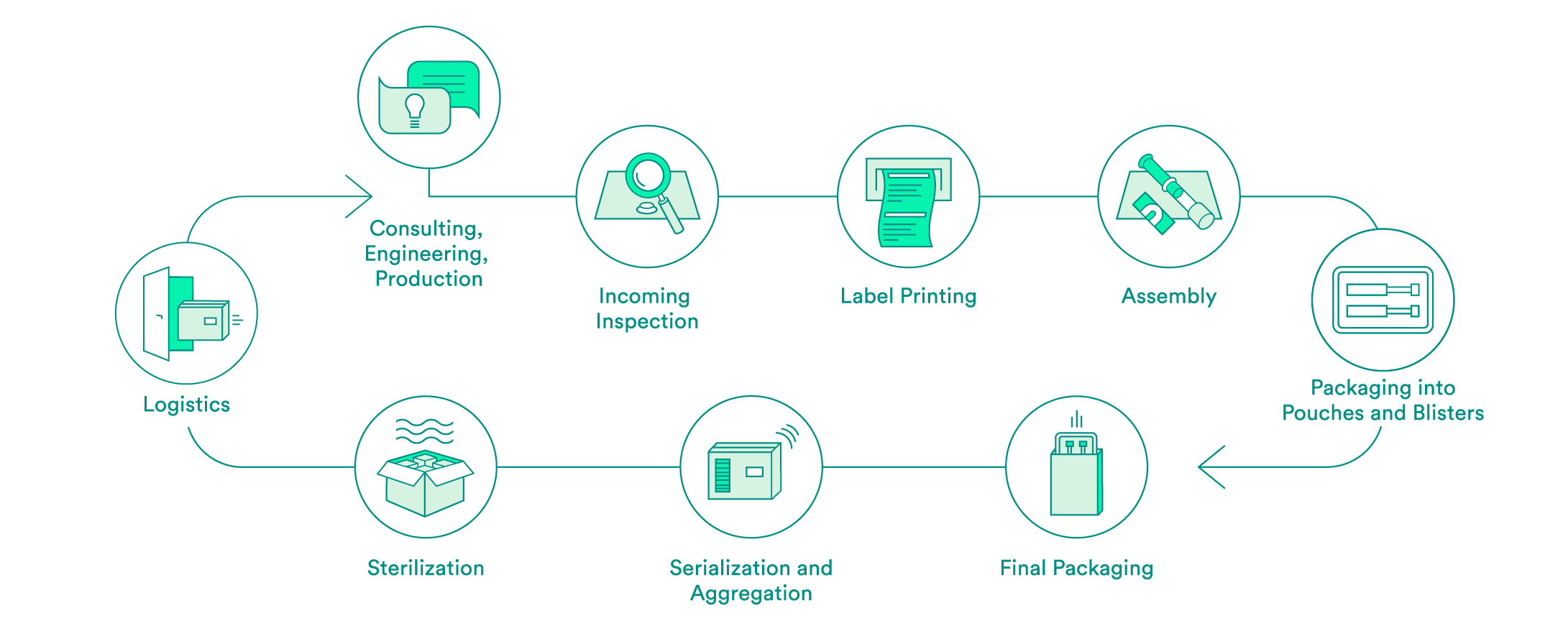
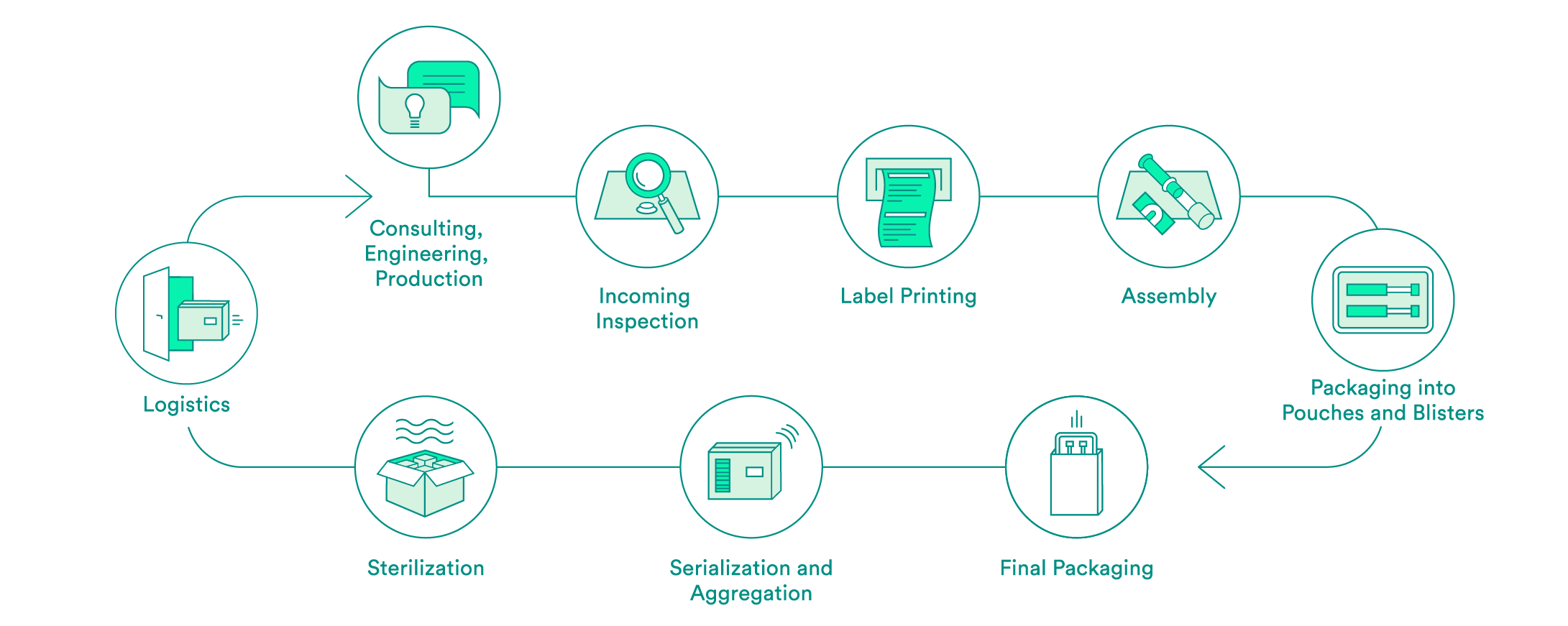
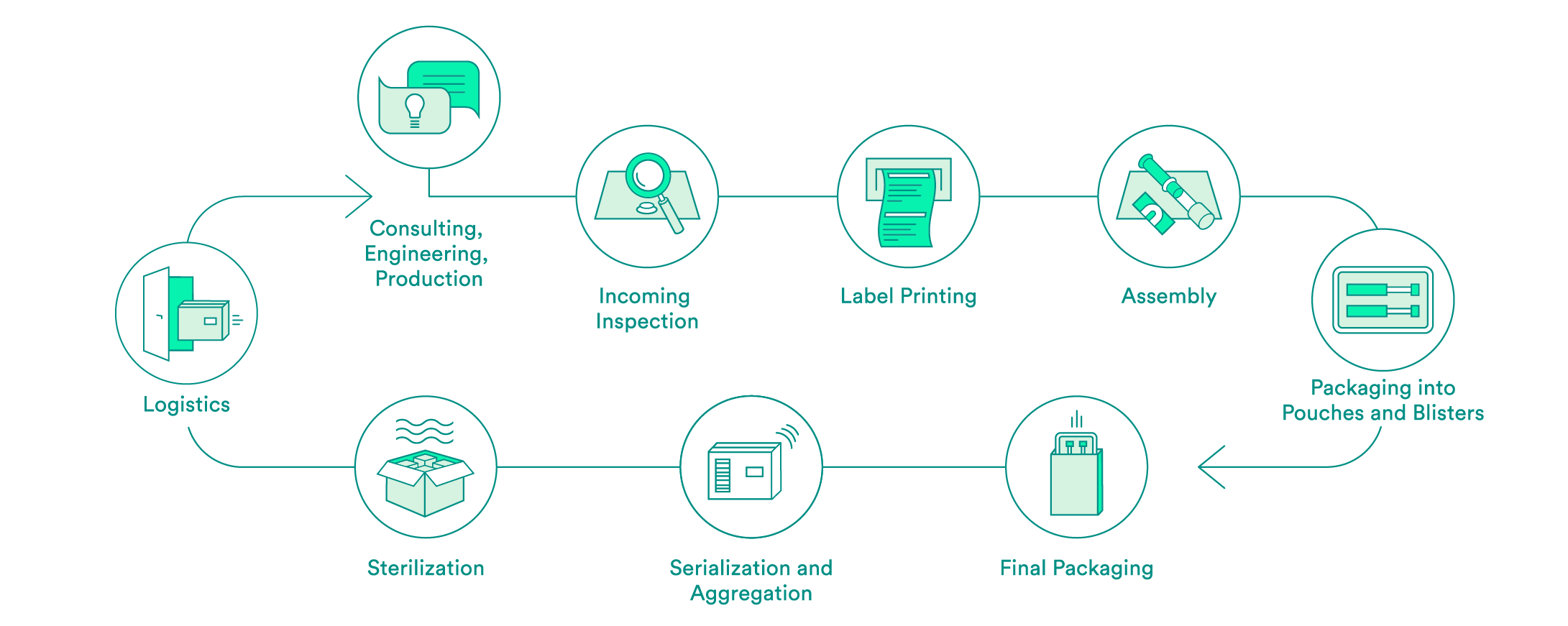
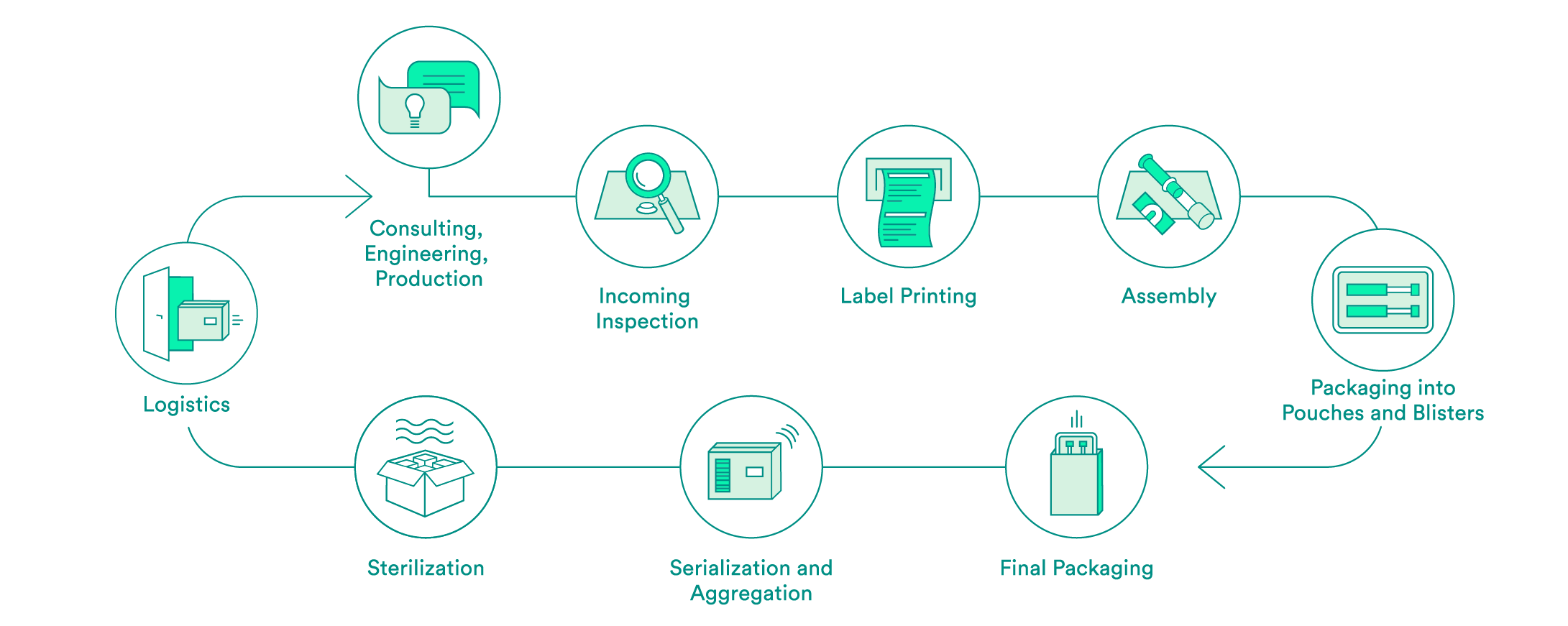
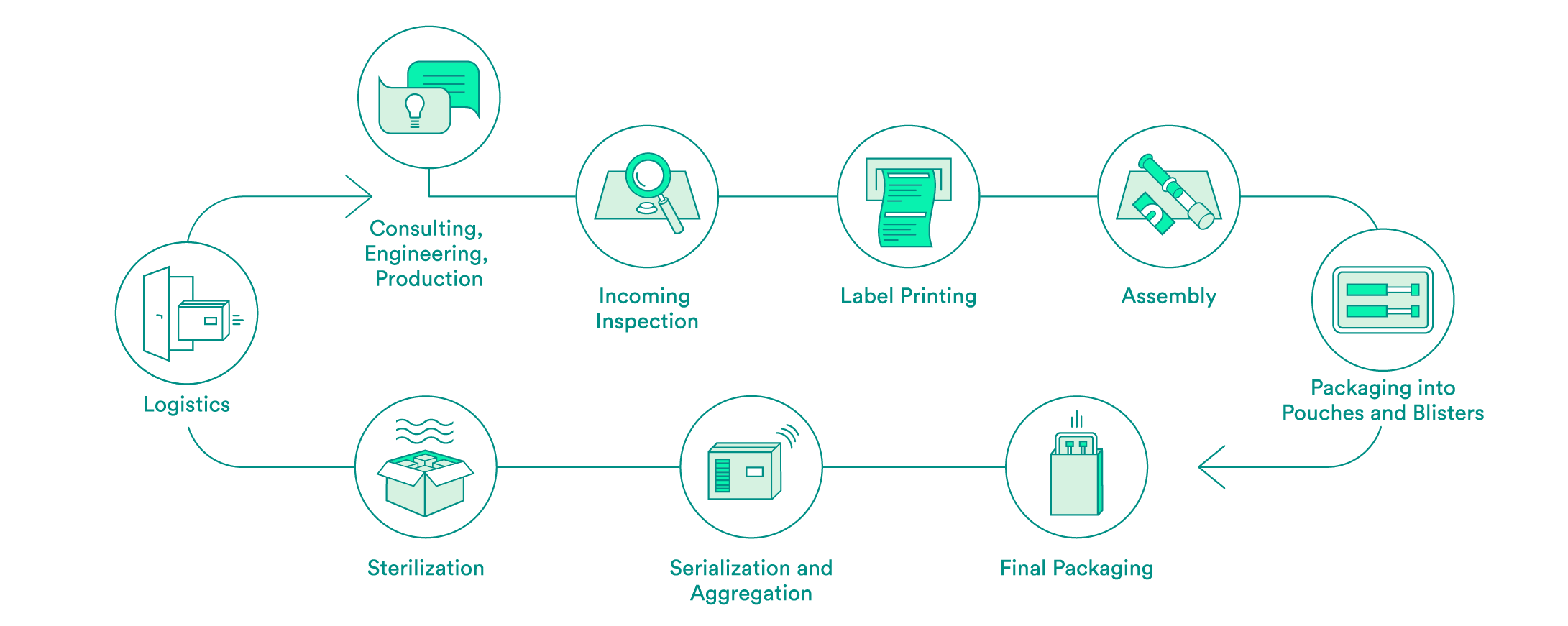
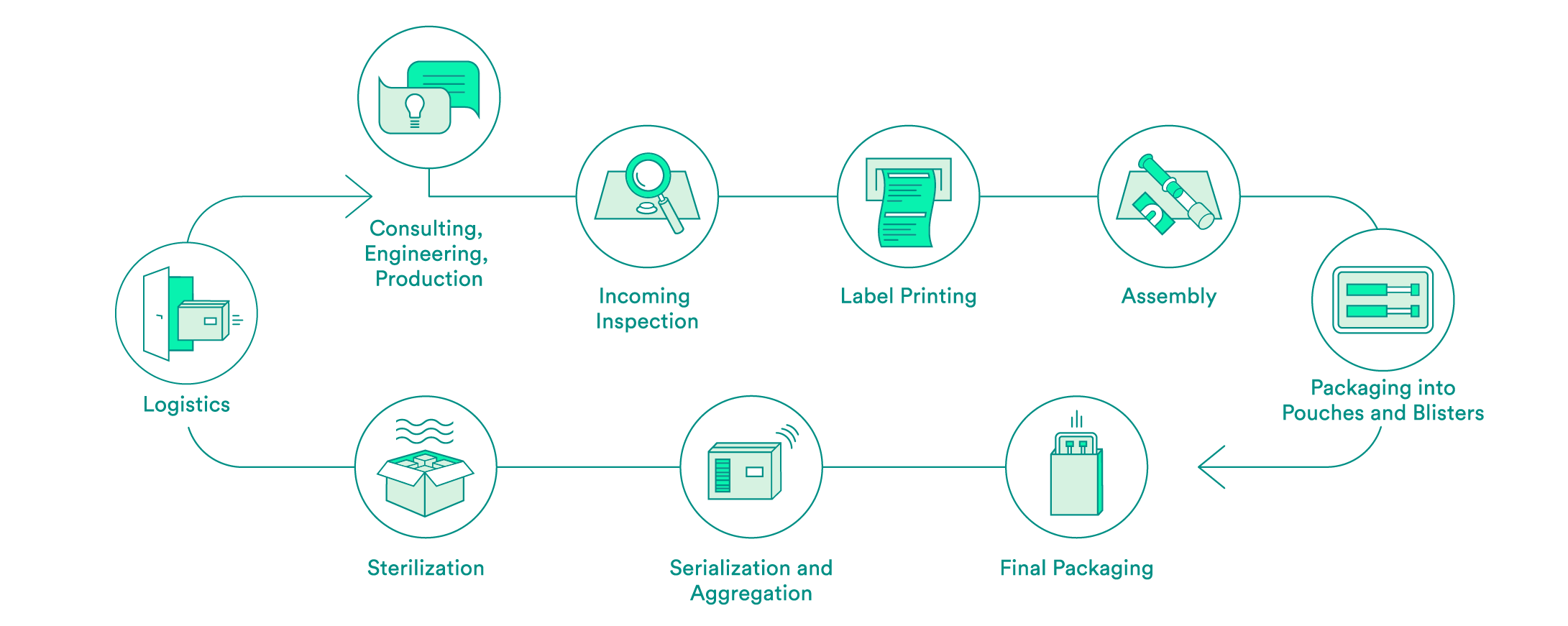
We take care of all contract packaging steps for our customers, from engineering secondary or tertiary packaging to the sterilization of the final packaging. We would be pleased to do so for your pharmaceutical products as well.
This is what you can expect from our contract packaging service for pharmaceutical products
- Packaging processes specialized for pharmaceutical products
- Knowledge of country- and market-specific regulations
- Modern infrastructure with a high level of automation
- Record fast throughput times
- High level of service
- On the market for 30 years
Pharmaceutical-specific knowledge and process steps for contract packaging
Each industry has different requirements in terms of packaging solutions. We are specialized in medical devices and pharmaceutical products. Depending on the requirements and in accordance with your needs, we offer:
- Incoming inspection with chemical product identification
- Quality assurance using the latest inspection systems
- Assembly of pre-filled syringes (PFS)
- Labeling of pre-filled syringes (PFS), vials or ampules
- Packaging of vials, ampules, cartridges, etc. in blisters or pouches
- Cartoning
- Serialization in accordance with FDA requirements for the US
- Clean room ISO class 7
- Packaging without UV rays
- Refrigerated logistics (2 – 8°C)
- Sterilization with partners
- Refrigerated storage and logistics
Record throughput time: packed in 8 to 10 days
Using specialized processes and modern infrastructure, we guarantee a fast throughout time – so your products reach the market quickly. The high level of automation of our systems and the integration of the flows of data and goods increases the efficiency. We ensure maximum quality using the latest inspection systems that monitor all work steps.
We support you every step of the way, from the first consultation to implementation of the packaging solution – both for products that have already been approved and for those products still in the approval process. Our expert interdisciplinary team will help you to choose the packaging material, select the production environment and define the correct processes, including the logistics.
Upon request, we will carry out the incoming inspection of the pharmaceutical products delivered in the primary packaging. In addition to a visual inspection, we also offer chemical product identification through a partner.
We will take care of the layout and the printing of your labels. Various substrates are available to you. A Früh database or a connection to your system can be used for data transmission.
If necessary, we can carry out assembly in clean room conditions in accordance with ISO class 7. The assembly of pre-filled syringes can be carried out manually, semi-automatically or fully automatically.
We have experts for the engineering and production of tailored secondary packaging. Blister packs can be tailored in-house to meet your needs or developed and produced from scratch. We can pack vials, ampules, cartridges and more in blisters and pouches. UV-sensitive products are packed in specially equipped rooms. If necessary, the products can also be packed under controlled conditions in an ISO class 7 clean room.
We carry out the final packaging process. We can pack products manually or semi-automatically in individual boxes – including the package insert. All components are checked to ensure that the correct product is in the package, and the variable data is printed on the box.
Upon request, we add a serial number to the packaging and aggregate deliveries at the box and/or the pallet level. For the US sales market, we guarantee that customers have serialization in accordance with FDA requirements.
We assist you with implementation of pilot tests and validations, and have reliable partners for the sterilization of your products.
We offer the option of cold storage (storage at 2 – 8°C) for your temperature-sensitive products, and we also have refrigerated logistics partners that can ensure transportation at these conditions.
For large orders, we offer door-to-door service: We pick your products up from you one to five times per week and then deliver them back to you in their packages.
For small orders, we recommend that you deliver and pick up your products from us yourself in order to keep the processing times as short as possible.
Do you have any questions about our contract packaging pharma? We will be happy to provide you with information.
Christian Reiser
Sales Manager & Key Account Manager
+41 58 258 24 08