Recyclable material with high barrier for pasteurization and sterilization applications
Discovering the power of mono-packaging
Mono-packaging material, a game-changer in terms of sustainability, refers to the use of a single type of material for packaging. This simplifies recycling processes and addresses the challenges posed by multi-material packaging. This approach contributes to a more efficient recycling process and aligns with global efforts to achieve recycling targets and create a more sustainable circular economy for packaging materials.
Unleashing sustainable solutions for ready meals
In the context of the global ready meals market, which was worth USD 135 billion in 2022 and is expected to reach USD 219.90 billion by 2032, the impact of sustainable practices could be groundbreaking. While they may not compete with carefully crafted meal plans, convenience foods offer significant benefits in our busy lives, including time savings, on-the-go convenience and high-quality, healthy options.
Following consumer trends
As consumer behavior merges with trends towards healthier lifestyles and greater sustainability, mono-material is proving to be a compelling solution for ready meals. We have teamed up with pioneers and invested in the development of mono-material films for for pasteurizable and acidic foods: This sterilization-resistant oxygen barrier system preserves food quality and color, making it a sustainable choice.
Discover here more about our film manufacturing.
Mastering Process and Packaging Validation – ISO 11607
Ensuring compliance with ISO 11607 is imperative in the field of packaging for terminally sterilized medical devices. The internationally harmonized standard is separated into two parts:
- ISO 11607 – 2 covers the requirements for the processes of forming, assembly and sealing of the packaging. The processes involved are known as a (packaging) process validation.
- ISO 11607 – 1 focuses on the requirements for the materials, sterile barrier system and packaging system. One of the main aspects of to be considered are the packaging system’s performance and stability. The processes involved are generally known as a packaging system design validation or simply packaging validation.
ISO 11607 – 2 – Process Validation
A process validation at a minimum must constitute of an installation qualification (IQ), an operational qualification (OQ) and a performance qualification (PQ). The general goal is to make sure that the equipment operates as intended and that the packaging can be produced as consistently, repeatably and reliably as expected.
Let’s take a closer look at the qualification phases:
- IQ – Installation Qualification
The purpose of Installation Qualification is to ensure that equipment, systems, and facilities are properly installed and meet the specified design criteria.
- OQ – Operational Qualification
OQ focuses on verifying and documenting that the equipment or system operates according to its operational specifications under normal operating conditions. For sealing processes this includes the determination of the lower process limits (LPL) and upper process limits (UPL) for critical process parameters like sealing temperature, time and pressure.
- PQ – Performance Qualification
This phase focuses on demonstrating that the equipment or system consistently performs according to its specified design and operational requirements in the intended operating environment. For sealing processes, packaging which were sealed at nominal process limits (NPL) are checked.
During the OQ and PQ, it is crucial to assess the effectiveness of the sealing through seal integrity testing, seal strength measurements and other packaging tests. See below for further information on a selection of these tests.
ISO 11607 – 1 – Material testing
Major parts of ISO 11607 – 1 focus on requirements for the materials used for the packaging system and sterile barrier systems, as well as for the packaging system and sterile barrier system themselves.
A selection of some of the most important requirements is listed in the following:
- Microbial barrier properties
Ensuring a microbial barrier is critical for ensuring integrity once sterilized, as it prevents the ingress of microorganisms.
- Biocompatibility
Biocompatibility should show that there are no critical substances that could migrate from the packaging material to the medical device and cause any harm for patients. Testing can involve cytotoxicity, nonvolatile residue etc.
- Physical and chemical properties
Conforming to a variety of physical and chemical properties ensures that the materials are suitable for the intended purpose of ensuring the overall product safety and especially ensuring the integrity even under physical stress during distribution. Testing for physical and chemical properties can for example involve burst testing, cleanliness, printing & coating, tensile properties, thickness etc. and have to conform to established properties and/or values.
- Sterilization compatibility
The materials used for the packaging system have to be compatible with the intended sterilization method (e.g., Gamma-ray, X‑Ray, EtO). Compatibility can for example be supported by testing of permeance, biocompatibility, seal strength and burst testing after exposure to the sterilization agent.
ISO 11607 – 1 – Packaging Validation
The main goal of a packaging validation is to establish evidence that the packaging system offers sufficient protection of the product, and, most importantly, that the sterile barrier system is capable of upholding its integrity after sterilization, distribution and storage.
A packaging system subjugated to a packaging validation should be manufactured under standard operating procedures using fully qualified equipment & sealing processes and sterilized using a validated sterilization process. In every aspect possible the worst-case scenario should be considered.
This includes:
- Worst-case medical device
If it is intended to use the same packaging system for a medical device product family (i.e., medical devices that are similar but not identical) the worst-case configuration out of this product family should be used. This can for example be the heaviest, bulkiest and/or sharpest configuration which poses the most stress on the packaging system.
- Worst-case sealing
Sterile barrier system (SBS) for the packaging validation should be sealed at LPL, which imposes the highest risk for compromising the integrity of the SBS.
- Worst-case sterilization
The samples for the packaging validation should be sterilized in a way to impose a higher challenge compared to the routine sterilization parameters. This for example can entitle a double-dose and/or a double-cycle sterilization.
A comprehensive packaging validation involves performance and stability testing. While it is not strictly prohibited to combine these two testing fields, it is recommended that they are kept separate from each other.
Packaging Validation – Performance Testing
Packaging performance testing involves a series of simulations and evaluations to ensure that the packaging system can withstand the challenges encountered during the expected handling and distribution.
Some important aspects to consider:
- Does the product ship as an individual box and/or as an unitized load on a pallet?
- Will the product ship as full truckload (FTL) or Less-Than-Truckload (LTL)
- In what climatic zones of the world will the product ship? What are the expected climatic conditions which could be encountered during transit?
- Will the product be shipped by boat, truck, rail and/or air?
Based on this information, it should be considered what the most appropriate consensus standard is that best aligns with the individual requirements.
Here is a selection of the most commonly used standards for packaging performance testing for medical device packaging:
- ASTM D4332
This standard is often used as a basis for testing environmental climatic conditions encountered during transit. Depending on which climatic zones the product will be shipped, the temperature can for example range from ‑40 °C to 70 °C and 10 % — 95% relative humidity. Knowledge of the expected climatic conditions during transit is essential, as these conditions should be tested in a series of simulated climatic conditioning steps which combined is known here at Früh as a climatic cycle.
- ASTM D4169
This widely used standard for transport simulations can be used as a basis to evaluate the ability of the packaging system to withstand the distribution environment with a focus on mechanical hazards, such as dropping, staking and vibration. Depending on the shipment configuration (single box vs. unitized load on a pallet) and expected shipment method (truck, rail, cargo ship and/or plane) the user can choose from a variety of different test sequences known as distribution cycles (DC).
- ISTA 3A
This standard shares many similarities with ASTM D4169 and is therefore another great option for transport simulations for medical device packaging. One important difference is that testing according to ISTA 3A can only be done on single boxes intended to be shipped via a parcel system. In general, testing according to this standard is more challenging but less customizable than its ASTM counterpart.
After performance testing, sterile barrier system integrity has to be proven through integrity testing. Seal strength measurements and other packaging tests can be included as well. See below for further information on a selection of these tests.
Packaging Validation – Stability Testing
The primary objective of stability testing is to assess the ability of the sterile barrier system to withstand the risks which can occur throughout the claimed shelf-life. The data from accelerated aging, which is aging conducted at elevated temperatures, is permissible to use for initial market launches, but the claimed shelf-life must be confirmed through real-time aging. ASTM F1980 is the go-to standard for establishing accelerated aging protocols for medical device packaging and gives guidance to calculate the time for the accelerated aging based on the Arrhenius equation.
Let’s take a look at an example:
An accelerated aging temperature of 55 °C, an assumed real-time aging temperature of 25 °C and an Arrhenius aging factor of Q10 = 2 allows for a reduction of the real-time aging time by a factor of 8. Therefore, for example, accelerated aging would shorten the time of 10 years real-time aging to 456.3 days (approximately 1.25 years).
After stability testing, sterile barrier system integrity has to be proven through integrity testing. Seal strength measurements and other packaging tests can be included as well. See below for further information on a selection of these tests.
Integrity and other packaging tests
Packaging systems including all sterile barrier systems have two very important functions in the life cycle of a terminally sterilized medical device:
- Protection of the medical device from damages until the point of use
- Ensuring sterility of the medical device until the point of use
Whether the packaging system is able to fulfill these functions has to be tested after each step of the packaging validation.
For this purpose, ISO 11607 – 1 lists a multitude of tests to choose from. Some of the most commonly used tests are listed below.
Integrity Tests:
- Bubble Emission Test through internal pressurization (ASTM F2096)
The packaging is placed underwater and subjected to increased internal pressurization. Constant bubble streams emerging from the packaging point to the existence of leaks.
- Bubble Emission Test – negative pressure (ASTM F3078)
The packaging is placed underwater in a chamber. A certain level of vacuum is applied to the chamber, which causes a pressure differential between the inside and outside of the package. Constant bubble streams emerging from the packaging point to the existence of leaks.
- Dye Penetration Test – porous packaging (ASTM F1929)
A dye solution is injected into the package, or, alternatively, the edges of each side of the seal are dipped into the dye solution. Each side of the seal seam is inspected for channels.
- Dye Penetration Test – non-porous packaging (ASTM F3039)
A dye solution is injected into the package. Each side of seal seam is then inspected for channels. Under certain conditions, it is additionally possible to inspect the flat surfaces of the packaging for leaks.
- Visual Inspection of the sealing (ASTM F1886/F1886M)
This method focuses on visually detecting channels and other quality relevant seal characteristic, such as undersealing, oversealing, folds etc.
Material Tests:
- Tensile Strength and Elongation at Break (DIN EN ISO 527−1÷527−3)
Tensile strength and elongation at break are two important mechanical properties used to characterize the behavior of materials under tension. Tensile strength is the maximum amount of tensile (stretching) stress that a material can withstand before failure or breaking. Elongation at break measures the extent to which a material can stretch or deform before it breaks. Both values are measured by pulling apart a strip of the test material.
- Puncture Resistance (DIN EN 14477 or ASTM F1306)
Puncture resistance is a mechanical property that measures a material’s ability to withstand the penetration of sharp objects without tearing or puncturing. The test is performed by forcing a probe into the material until complete penetration, resulting in the measurement of the force and energy at break.
Other packaging tests:
- Seal strength (ASTM F88/F88M)
The purpose of seal strength testing is to evaluate the ability of a seal to withstand the stress and pressure encountered during transportation, handling, and storage. For this test, a defined section of the seal seam is pulled apart until full separation, resulting in the measurement of the seal strength.
- Visual inspection of packaging (in-house method)
By visually examining packaging signs of damage, such as tears, punctures, or deformation, weaknesses and vulnerabilities in the packaging system can be identified. Investigating these issues helps to prevent product loss, contamination, or damage, ultimately safeguarding the sterile barrier system and the packaged product.
- Dimensional measurements (ASTM F2203 or in-house method)
Dimensional measurements can be taken from any part of the packaging. Most commonly this test is used to measure the seal width of the sterile barrier system either with a steel ruler (ASTM F2203) or with a digital caliper (in-house method).
Gold status for stand construction at Pharmapack
We were awarded Gold status for our sustainable stand construction at Pharmapack. Thanks to the multiple use of the graphic wall, the screen, the rental furniture and the clever power supply planning, the event3 stand construction team was able to fulfill all sustainability requirements perfectly.
Quality standards
Our equipment is qualified, and our processes and packaging solutions are validated. At Früh Verpackungstechnik AG, we are committed to complying with Good Manufacturing Practice (GMP) standards, which form the basis for high-quality production processes in the pharmaceutical and medical device industries.
We subject our qualified facilities and environmental conditions as well as all production processes to a continuous validity check with the following GMP standards:
Design qualification (DQ)
In doing so, we ensure that the planning and design of our facilities meet the defined requirements from the outset. This is the first step in guaranteeing the quality and functionality of our solutions.
Installation qualification (IQ)
During installation qualification, we check that our facilities and devices are correctly installed and configured. This check ensures that all components function in accordance with the manufacturer’s specifications and our own high standards.
Design of experiment (DOE)
With this approach, we systematically examine the influence of various factors on our processes. This helps us identify optimization opportunities and increase the efficiency of our processes.
Operational qualification (OQ)
Operational qualification is a critical step in which we test the performance of our systems under defined conditions. We ensure that our facilities operate consistently and reliably under all intended operating conditions.
Performance qualification (PQ)
In this phase, we validate the performance of our processes in regular operation. We confirm that our systems will operate consistently and in accordance with the specified specifications over a longer period of time.
Through these comprehensive qualifications based on GMP, we ensure that our facilities and processes not only meet the highest standards but are also regularly checked and updated. This continuous monitoring and improvement is part of our commitment to providing our customers with the highest quality and safety in medical device and pharmaceutical packaging.
We subject our qualified systems and environmental conditions as well as all production processes to continuous validation checks.
Validation
We understand the importance of comprehensive validation processes to ensure the quality and safety of our packaging solutions for the medical devices and pharmaceutical industries. In doing so, we comply with the mandatory requirements for the GPA guidelines of Swissmedic. We offer various types of validations that are tailored to the needs of our customers:
Hygiene validation
Thanks to our many years of expertise in packaging pharmaceutical and medical devices under controlled clean room conditions according to ISO 7 and ISO 8 as well as the continuous improvement of our environmental monitoring system, we ensure the highest safety standards for the products we package. We are committed to providing the best possible protection against external particulate and microbiological contamination for our customers’ products.
Process validation
The effectiveness of a process always depends on the quality of its individual components. We therefore pay particular attention to the qualification of all key processes (e.g. the sealing process) as well as our clean rooms and packaging machines. This careful preparatory work forms the basis for the actual validation of the product packaging process.
Product validation
Our focus is on confirming all requirements of the products and, in particular, the individual customer requirements for the products to be packaged. In addition to the qualification of the process components, this validation also includes stability and performance tests such as packaging integrity tests. Our goal is to ensure the highest level of product and process safety for the packaged customer products.
Sterilization validation
Products subjected to final sterilization (e.g., gamma or X‑ray radiation) undergo sterilization validation in accordance with the EN ISO 11137 series of standards in cooperation with our customers. This validation ensures that the normative requirements for the reduction of microbial contamination are met to mark a medical device as ‘STERILE’.
Our comprehensive validation processes ensure the quality and safety of our packaging solutions.
MD&M West and Pharmapack 2024
We will be exhibiting almost simultaneously on 2 continents: On January 24 and 25, you can meet our team at Pharmapack in Paris, booth J89. From February 5 to 8, we will be exhibiting as a partner of Swiss Global Enterprise at MD&M West in Anaheim, booth 2956.
Energy conservation: not an empty promise
Facts and figures
In the spring of 2022, an energy and raw materials crisis began in Europe the likes of which we have not seen in the last two decades. The changes in the market hit the packaging industry with full force and changed the entire industry. Depending on the country, the cost of energy in Europe rose to dimensions that are new to all of us.
Früh AG has been a leader for years in the packaging sectors of medical technology, pharmaceutical industry as well as in the chemical-technical sector. Our packaging materials and the associated technologies are very energy-intensive and thanks to the fact that we set the course towards energy efficiency years ago, we were well positioned against the energy crisis of 2022. In addition to our contribution to voluntary energy efficiency, which we implement with the Energy Agency for Industry (EnAW), we work with the Ecovadis organization — with silver status.
We achieved the major and for us important effect for energy consumption with the new construction of our packaging production. We have invested in state-of-the-art infrastructure and production facilities at our Fehraltorf site in order to ensure, on the one hand, the increasing demand for our products, the competitiveness as well as the sustainability of our production. For example, we work with the most modern printing and laminating machines and a new RNV plant (regenerative thermal afterburning). Whereas total energy consumption in 2014 was 6 million kWh, in 2022 it was 10 million kWh. At the same time, we doubled our production output during this period.
This results in a reduction of 20% in gas consumption despite a massive increase in production. In the case of electricity, where we obtain 95% from sustainable production, we were able to reduce consumption by over 10%. If we compare the energy consumption in the medical technology area in relation to the packaged implants, we have even been able to achieve a reduction of 50% since 2014.
Another key figure is the kWh consumption per employee: In 2014, this was approx. 20,000 kWh, currently approx. 14,500 kWh, which corresponds to an improvement of almost 40%. The number of employees was 300 in 2014 and currently around 700.
By investing in forward-looking and efficient production lines, we have succeeded in massively reducing our CO2 emissions. The path to sustainability is a central topic at Früh Verpackungstechnik AG, which will also accompany us in the future.
If you are interested in how the above-mentioned reductions have been achieved in detail, we will of course be happy to provide you with figures and further information.
Conferences 2023
After the intensive trade fair spring, the next round starts soon. We will be exhibiting at these two conferences:
Oct 05 — 06 PPL Berlin (Pharmapackaging and Labeling).
Oct 09 — 10 WICD London (Wearable Injectors and Connected Devices)
Are you also attending? Take the opportunity and discuss your concerns and challenges with our sales men, together we will find a suitable solution for all requirements.
Accredited lab services
Our laboratory services are now accredited according to ISO/IEC 17025
For our customers, this means accredited test methods for:
- Integrity tests
- Bubble emission tests by internal pressurization or in a vacuum chamber
- Dye penetration tests for porous and non-porous packaging
- Visual inspection of seal seams
- Packaging and material tests
- Seal strength
- Tensile strength and nominal elongation at break
- Linear dimensions of packaging
- Puncture resistance
- Visual inspection of packaging
- Climatic conditioning and packaging validations
- Stability tests
- Performance tests
Please do not hesitate to contact us for individual consultation or further information.
MD&M East 2023
The première at MD&M East was a success! Yasin Gencel and Miki Velickovski were able to inspire the many new interested parties with our products and also inform about the opening of the site in Exton PA.
Connect in Pharma 2023
The première also went well in Geneva at «Connect in Pharma 2023». Chris Reiser, Ivan Orlando & Markus Bischof provided information primarily about pharmaceutical packaging solutions.

Medtec2023
Medtec 2023 was inaugurated with a new booth design. Furthermore, the focus was on the validated (and soon to be accredited) lab services.

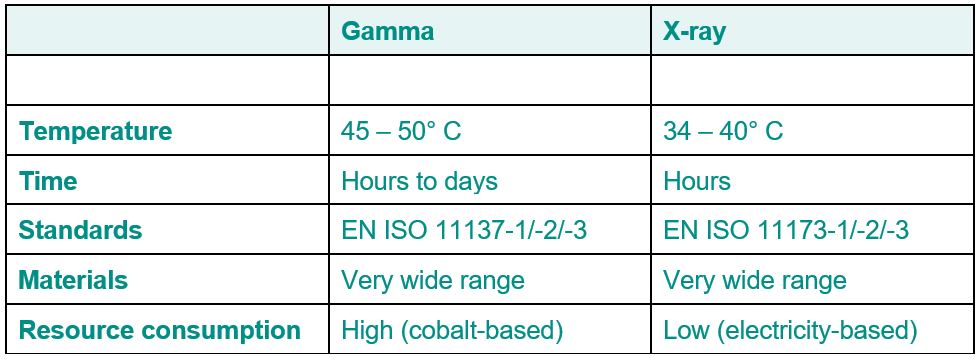
X-ray irradiation
The future-oriented alternative to gamma irradiation of medical devices
Gamma irradiation has been the usual method for sterilizing medical products for more than five dec-ades. However, there have been problems sourcing cobalt of late, calling into question whether this process is sustainable and efficient. X-ray irradiation has turned out to be a promising alternative, as it is a more environmentally friendly and cost-effective method that is accepted by the regulatory authorities and offers a number of advantages over gamma irradiation. Find out more about the chal-lenges of gamma irradiation and the compelling reasons for switching to X-ray irradiation in this arti-cle.
Uncertainty concerning cobalt sourcing for gamma sterilization
Gamma irradiation of medical products has been the state-of-the-art method for more than five dec-ades. Many products are treated with a minimum dose of 25 kGy in accordance with the validation process as per the EN ISO 11137-1/-2 standard and VDmax25 method. Gamma irradiation is defined as a clean and cold form of sterilization because no chemicals are used and the sterilization tempera-ture rarely exceeds 45° C. However, there have been increasing problems sourcing cobalt in recent years. Cobalt is used as a source of irradiation in most gamma equipment in the form of the radioac-tive isotope cobalt-60. Mining, processing and transforming cobalt-59 iron ore into cobalt-60 is a complex and expensive process. Furthermore, cobalt is becoming increasingly scarce. This is putting greater pressure on source operators, as cobalt-60 has a half-life of 5.27 years and cobalt sources must be replenished periodically in order to maintain activity and thus the strength of the source at the highest level possible. If the source strengthen wanes, this increases the irradiation duration for the medical product and results in backlogs if the source is not replenished in time. In addition, there is an increasingly critical view of transporting radioactive materials by land and by sea, resulting in in-creasingly stringent regulations. This has led to higher costs for source operators. Because of the controlled end storage of radioactive materials, gamma irradiation can be described as a clean tech-nology, but not a green one.
The environmental and economical alternative: Advantages of X-ray irradiation
One solution to the problem is X-ray irradiation, which has become increasingly popular in recent years. X-ray irradiation, also known as "bremsstrahlung," has the same basic effects as gamma irradi-ation on microbiological systems like bacteria, molds and yeasts. For this reason, X-ray irradiation is described in EN ISO 11137-1/-2 as equivalent to gamma irradiation and the validation requirements are exactly the same. However, X-ray irradiation has considerable advantages. For example, X-rays are produced by transforming or slowing accelerated electrons, while electrons are produced in a ma-chine by means of an electromagnetic field. The electrons are then accelerated to an energy level of up to 7 MeV. The X-ray machine works solely on the basis of electricity. Thus, there are no costs for cobalt and no transportation costs, as is the case with gamma irradiation. As with gamma irradiation, absolutely no chemicals are used in X-ray irradiation. Since there is also no necessity to dispose of waste or other materials, X-ray irradiation can be classified as a clean and green technology.
Why X-ray irradiation is the preferred method for sterilizing medical products
There are two other advantages. The first is that while X-rays have the same properties as gamma rays, their lower average level of energy means they do not penetrate materials as deeply as gamma rays do and thus represent a lower risk of structural modifications, especially in plastics. The second advantage is that there is a much lower thermal load on the products than is the case with gamma irradiation because the products are only exposed to the rays for a few seconds in total. As a result, the temperature does not climb as high as it does with products that are kept in a gamma irradiation room for several hours. For these reasons, it can be said that X-ray irradiation is the preferred method for treating medical products effectively and sustainably. X-ray irradiation is a clean and green tech-nology that is carried out under the same regulatory requirements as gamma irradiation and has the same effects on microbiological systems. X-ray irradiation is softer and produces a lower thermal load than gamma irradiation, which reduces the risk of transforming the product's characteristics. In addition, the operator of the irradiation equipment does not need to worry about sourcing cobalt and customers do not need to worry that equipment capacity will fall. X-ray irradiation is the method of the future and an increasing number of medical products will make the switch in the coming years from old methods like gamma and ethylene oxide to X-rays.
Did you know that we need less than 48 hours for sterilization and transport to you? And refrigerated goods only require 24 hours for Swiss customers.
If you have any further questions about the sterilization options, please contact our specialist Alexander Müller.
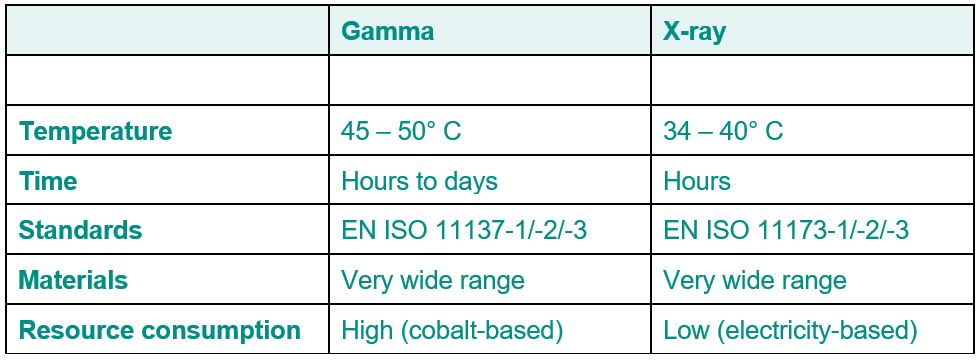
X-ray irradiation
The future-oriented alternative to gamma irradiation of medical devices
Gamma irradiation has been the usual method for sterilizing medical products for more than five dec-ades. However, there have been problems sourcing cobalt of late, calling into question whether this process is sustainable and efficient. X-ray irradiation has turned out to be a promising alternative, as it is a more environmentally friendly and cost-effective method that is accepted by the regulatory authorities and offers a number of advantages over gamma irradiation. Find out more about the chal-lenges of gamma irradiation and the compelling reasons for switching to X-ray irradiation in this arti-cle.
Uncertainty concerning cobalt sourcing for gamma sterilization
Gamma irradiation of medical products has been the state-of-the-art method for more than five dec-ades. Many products are treated with a minimum dose of 25 kGy in accordance with the validation process as per the EN ISO 11137-1/-2 standard and VDmax25 method. Gamma irradiation is defined as a clean and cold form of sterilization because no chemicals are used and the sterilization tempera-ture rarely exceeds 45° C. However, there have been increasing problems sourcing cobalt in recent years. Cobalt is used as a source of irradiation in most gamma equipment in the form of the radioac-tive isotope cobalt-60. Mining, processing and transforming cobalt-59 iron ore into cobalt-60 is a complex and expensive process. Furthermore, cobalt is becoming increasingly scarce. This is putting greater pressure on source operators, as cobalt-60 has a half-life of 5.27 years and cobalt sources must be replenished periodically in order to maintain activity and thus the strength of the source at the highest level possible. If the source strengthen wanes, this increases the irradiation duration for the medical product and results in backlogs if the source is not replenished in time. In addition, there is an increasingly critical view of transporting radioactive materials by land and by sea, resulting in in-creasingly stringent regulations. This has led to higher costs for source operators. Because of the controlled end storage of radioactive materials, gamma irradiation can be described as a clean tech-nology, but not a green one.
The environmental and economical alternative: Advantages of X-ray irradiation
One solution to the problem is X-ray irradiation, which has become increasingly popular in recent years. X-ray irradiation, also known as "bremsstrahlung," has the same basic effects as gamma irradi-ation on microbiological systems like bacteria, molds and yeasts. For this reason, X-ray irradiation is described in EN ISO 11137-1/-2 as equivalent to gamma irradiation and the validation requirements are exactly the same. However, X-ray irradiation has considerable advantages. For example, X-rays are produced by transforming or slowing accelerated electrons, while electrons are produced in a ma-chine by means of an electromagnetic field. The electrons are then accelerated to an energy level of up to 7 MeV. The X-ray machine works solely on the basis of electricity. Thus, there are no costs for cobalt and no transportation costs, as is the case with gamma irradiation. As with gamma irradiation, absolutely no chemicals are used in X-ray irradiation. Since there is also no necessity to dispose of waste or other materials, X-ray irradiation can be classified as a clean and green technology.
Why X-ray irradiation is the preferred method for sterilizing medical products
There are two other advantages. The first is that while X-rays have the same properties as gamma rays, their lower average level of energy means they do not penetrate materials as deeply as gamma rays do and thus represent a lower risk of structural modifications, especially in plastics. The second advantage is that there is a much lower thermal load on the products than is the case with gamma irradiation because the products are only exposed to the rays for a few seconds in total. As a result, the temperature does not climb as high as it does with products that are kept in a gamma irradiation room for several hours. For these reasons, it can be said that X-ray irradiation is the preferred method for treating medical products effectively and sustainably. X-ray irradiation is a clean and green tech-nology that is carried out under the same regulatory requirements as gamma irradiation and has the same effects on microbiological systems. X-ray irradiation is softer and produces a lower thermal load than gamma irradiation, which reduces the risk of transforming the product's characteristics. In addition, the operator of the irradiation equipment does not need to worry about sourcing cobalt and customers do not need to worry that equipment capacity will fall. X-ray irradiation is the method of the future and an increasing number of medical products will make the switch in the coming years from old methods like gamma and ethylene oxide to X-rays.
Did you know that we need less than 48 hours for sterilization and transport to you? And refrigerated goods only require 24 hours for Swiss customers.
If you have any further questions about the sterilization options, please contact our specialist Alexander Müller.
Exhibitions 2023
Things are heating up at Früh in May and June, as we are participating in these three great trade shows:
May 23 – 25 in Nuremberg at Medtec, Hall 3c, booth 15
June 13 to 15 in New York at the MD&M East, booth 1557
June 14 – 15 in Geneva at Connect in Pharma, booth D3
We can hardly wait & are already looking forward to many visitors!
Pharmapack 2023
Preparations for the first trade show in 2023 are in full swing: At the beginning of February, we will present our new congress booth at Pharmapack in Paris (booth G88).
Visit us at G88 — our sales team is looking to seeing you there!
First production site in the USA
We are delighted to inform you that we will be opening our new production site in Exton (PA). From the end of 2023, “Fruh Packaging Inc.”, which will operate as an independent company, will meet its clients’ needs by expanding its packaging services to the USA.
After careful consideration, we have decided to enter America’s east coast market for contract packaging for medical devices. The service catalog includes the same options as those offered in Switzerland. This will allow us to offer the valued expertise our clients have come to expect from us, just 40 minutes from Philadelphia.
The infrastructure qualification and certification phase will begin in October 2023, with plans to start production by Q1/2024. We will remain an independent, family-run business, and will be represented in Exton by Alexander Früh.
We are already looking forward to this exciting milestone and will, of course, keep you up to date with how the project is progressing.
Sustainable practices
The topics of “Energy + Sustainability” have not only been on our minds since the current crisis, but have been part of our strategic actions for a long time and serve as a basis for investments in sustainable and efficient technologies.
Would you like to know what/how much e.g. the optimization of the compressed air network, weaker logo lighting or regenerative thermal oxidation cause? Read about it here.
Print data portal
In order to guarantee error-free processing of projects, we use a cloud-based print data portal, which is also operated by the customer and his agency. The tool is used for efficient communication and approvals and enables error-free processing including quality assurance and archiving. The automated log book shows each individual process step and the associated responsible person. This way the correspondence can be reduced to the essentials and everyone knows exactly what has been done in advance or which step is next.
The release cycle (draft to GfP) takes between 2 to 4 weeks, depending on the product and customer.
Exhibition: PDA in Palm Springs
Visit us on October 19 and 20 at the PDA in Palm Springs (booth 708). The focus is on “contract packaging pharmaceuticals”, but of course we are also happy to show you other packaging solutions.
Implant
The contractpackaging for medical devices goes through 9 steps in a record processing time of 8 to 10 days.
1) consulting, engineering, and production for customized primary, secondary and tertiary packaging
2) incoming inspection with quantity and product control
3) clean room label printing in germ-free environment
4) final rinsing in clean room according to ISO class 7
5) assembly as requested in clean room conditions
6) clean room packaging in inert gas atmosphere
7) final packaging and cartoning
8) sterilization with gamma rays, X‑rays, or steam
9) logistics with “door-to-door” service
Record processing time in contractpackaging
Record processing time in contractpackaging medical: from incoming inspection to dispatch in 8 to 10 days including sterilization.
Made possible by specialized processes and modern infrastructure from a single source.
Peel pouches configurator
Peel pouches offer reliable protection as primary, secondary and tertiary packaging for medical devices and pharmaceutical products. Configure your custom-made peel pouch here.
Exhibition: PDA Basel
At the PDA in Basel, the focus is on advanced packaging solutions. Visit us on June 2 and 3 at the congress center Basel, we will be happy to advise you at booth 23.
Ecovadis: Silver
We are proud to have achieved “Silver” status with EcoVadis in the areas of environment, labor & human rights, ethics as well as sustainable procurement.
Exhibition: Medtec in Stuttgart
At Medtec, which takes place from 3 to 5 May in Stuttgart, the entire sales team of FRÜH awaits you. For the very first time, you have the opportunity to take a peek at our production facilities thanks to our virtual reality glasses.
You will find us at booth 10 – 212, pay attention to the floating FRÜH logo, it cannot to be overlooked.
For further information click here.
Exhibition: Pharmapack in Paris
On 18 and 19 May, Pharmapack will take place in Paris, it’s the European hub for the pharmaceutical packaging and drug delivery device industry. During these two days, we will be happy to show you how we handle the requirements for pharmaceutical contract packaging and which solutions are already available.
Our stand number is G88 — Bienvenue!
For further information click here.
Exhibition: MD&M in Anaheim
From 12 to 14 April, we will present ourselves as a partner of Swiss Global Enterprise in the Swiss Pavilion at the MD&M in Anaheim. The focus is on packaging materials for medical and technological and pharmaceutical products as well as for contract packaging solutions for medical technology and pharmaceuticals.
Visit us at booth #2869 in the Conventions Center in Anaheim – we look forward to seeing you!
For further information click here.
Exhibition: Pharmapack in Paris
We will be exhibiting at Pharmapack in Paris on October 13 and 14.
Whether you are looking for a primary packaging for enteral or parenteral use, a packaging for medical technology products such as implants or a packaging solution for syringes and vials, we have the right packaging and will be happy to advise you at our booth G96. We are looking forward to your visit.
Find more information about the exhibition here.
Webinar: Aseptic Fill / Finish - A Holistic Approach
The demand for having a fast time to market on a high level of Quality and Yield has increased within the last years.
Our Webinar on the September 9 will give you an overview of the challenges and of Fill/Finish including sterilization and how to overcome them by working with SME’s from different companies and achieving a fast and reliable solution for bringing your product fast to the market.
Register here for our webinar.
https://www.business-review-webinars.com/webinar/Pharma/Aseptic_Fill__Finish_A_Holistic_Approach-LXcP8myF
Exhibition: MD&M Anaheim
From August 10 – 12 we will participate in the MD&M in Anaheim as exhibitor together with Switzerland Global Enterprise. After more than a year without participating in a trade fair, we are allowed to be there in person. With our team of experts we will advise you on packaging materials for medical and pharmaceutical products as well as contract packaging solutions for medical technology and pharmaceuticals. We are looking forward to seeing you at booth #2869 in the Conventions Center in Anaheim.
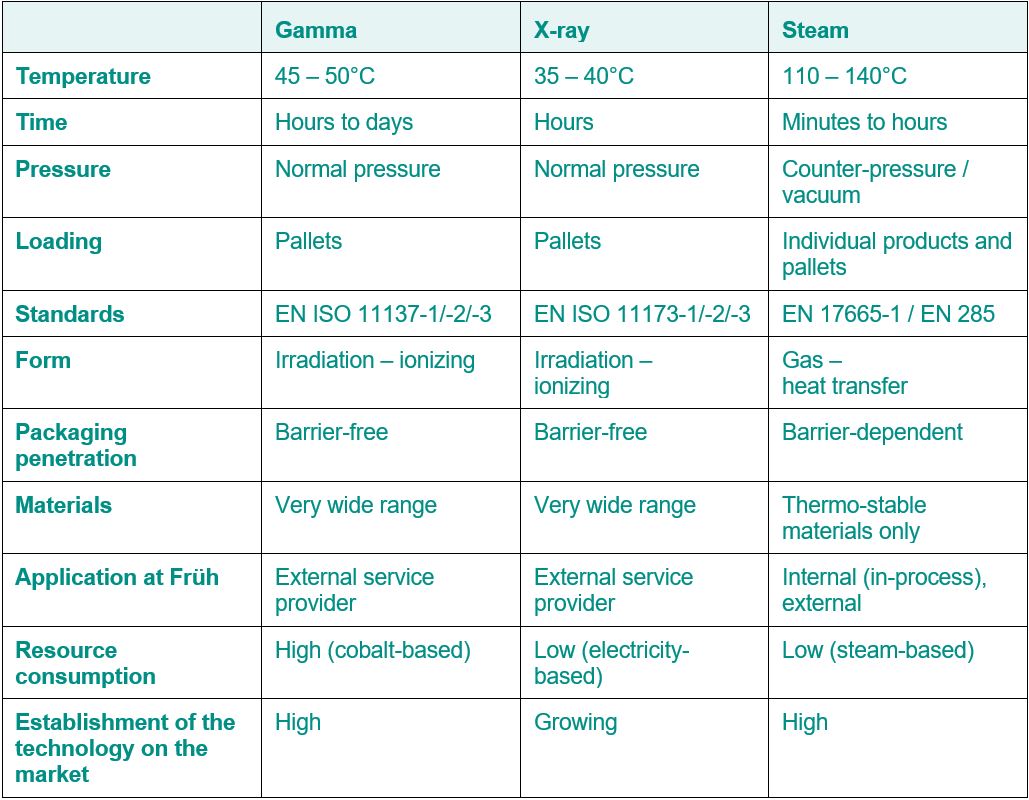
Gamma, X-Ray and Steam Sterilization
Are you developing a new packaging concept and would like to know which is the most suitable sterilization type? Or would you simply like an overview of the various types of sterilization? Below you will find an overview of gamma, x-ray and steam sterilization at Früh Verpackungstechnik AG.
Gamma sterilization
More than 40% of all medical products marked «sterile» involve the use of gamma irradiation. With this process, the gamma irradiation is released from the enriched cobalt-60 isotope. During beta decay, unstable cobalt-60 is converted into an excited state of nickel-60 and is then changed into stable nickel-60 through the emission of two gamma rays of 1.17 and 1.33 mega-electron volts. The half-life of this decay cascade is 5.27 years, making it the longest-lasting of all unstable cobalt isotopes.
Because of its characteristics, the gamma quanta can be described as both particles and as waves. This quality allows the gamma irradiation to penetrate deeply into the materials, which in turn makes it very well suited for treating medical products on, for example, EU pallets.
In most cases, medical products are treated to reduce and kill microorganisms, such as bacteria, that are on the products being treated. Medical products are said to be sterilized when the treatment process meets the requirements of EN 556-1. The procedure for sterilizing medical products using gamma irradiation is describe in standards series EN ISO 11137.
X-ray sterilization
Since the beginning of the 21st century, the market for medical products has seen another very effective sterilization type, so-called x-ray irradiation. As the name implies, this involves the use of x-rays or bremsstrahlung. The characteristics of this type of irradiation are very similar to gamma irradiation, but with the major difference that x-rays can be created artificially using electron acceleration and abrupt braking. With the help of a powerful electron accelerator of up to 7 MeV, electrons are accelerated to nearly light speed and shot at a target. In addition to a large amount of heat, the abrupt braking of the electrons also releases x-rays. These are then used, like gamma quanta, to treat medical products, for example, on EU pallets.
It can be said that x-rays penetrate materials better than gamma rays. This is because of the spectrum of rays that are produced, which will not be explained in further detail here. In terms of standards, there is no difference between the sterilization of medical products using gamma or x-ray irradiation
Steam sterilization
Steam sterilization is one of the oldest types of sterilization and is based on the principle of killing viable organisms and cells using heat. This results in the denaturation of proteins and the interruption of essential cell processes, which in turn leads to the death of microorganisms and viable cells.
In addition to medical products, a number of pharmaceutical products are treated using steam sterilization to reduce and kill microorganisms and other harmful organisms.
Tailored to the large range of products, there are a number of different processes available. These can be roughly divided into hot air sterilization and steam sterilization. Steam sterilization will be described in more detail here. In this process, the feed water, following the steam saturation curve, is heated in a steam sterilizer using a steam generator and released into the sterilization chamber as saturated steam. The chamber must first be emptied of air (evacuated) so the steam can reach the products to be sterilized without hindrance. Any remaining air would function as insulation during the steam sterilization and the steam might not be able to transfer all of its energy to the goods being sterilized. In addition, care needs to be taken that suitable packaging materials are used during steam sterilization through which the steam can pass and end up on the product being sterilized. In many cases, a fractionated pre-vacuum process is used for the sterilization of medical products, with a final vacuum drying process (FRVV-VMT, or fractionated pre-vacuum process – vacuum with drying). However, the process can also be adjusted in a modular manner depending on the product or process specification. Standards series DIN 58950 provides further details (pharmaceutical sterilization products) about the individual processes using heat and steam.
Material suitability - which type of sterilization is the right one?
In general, the same materials are suitable for irradiation using gamma rays and x-rays, as both of these sterilization types involve electromagnetic irradiation that have an ionizing effect on the irradiated material through the interaction of photons and electrons An overview of suitable materials for gamma ray, x-ray and steam sterilization can be found in AAMI TIR17:2017.
Please note that the use of x-rays, which are created through the acceleration of electrons using more than 5 mega-electron volts of energy, requires a material activation test. This requirement is defined in EN ISO 11137-1.
Comparison of the types of sterilization
Exhibition: Meet the Expert and Orthomanufacture
Meet the Expert will be held this year on April 28 and 29 in collaboration with Orthomanufacture. This will combine two congresses of the implant industry.
The scheduled presentations will now all take place digitally. In addition to the presentations, attendees will be able to meet in virtual rooms. We are pleased to be included with a presentation on microbiology and sterilization given by Peter Huonker, Head of Quality.
Virtual Showroom
Unfortunately, due to the current situation, no trade fairs can take place live on site, so we offer you an alternative. Switzerland Global Enterprise has launched a virtual trade fair of the Swiss packaging, processing and plastics industry.
We are pleased to be part of it. Visit us at our virtual booth.
s‑ge.com/showroom-frueh
Webinar - Peel pouch: specifications and application
The demands on packaging in the medical and pharmaceutical industry have increased. Much more extensive documentation is required today, especially with regard to validation and qualification in the production and use of packaging materials.
In our webinar on February 11, we will give you an overview of our peel pouches. We would like to show you which different materials are used and what the quality characteristics of our peel pouches are. We will show you the properties and specifications of the different material composites and how they can be used for your products.
Merry Christmas
We wish you a Merry Christmas and a happy and healthy New Year.
Exhibition: Pack Expo Connects 2020
Pack Expo Connects 2020 is a web-based event complete with live chats, live product demos and equipment and engaging educational opportunities, this will be the place to interact with packaging and processing professionals this year. It will take place on November 09 until November 13, 2020.
We are inviting you to attend Pack Expo Connects 2020. You can register for free.
You find more information here.
We are looking forward meeting you in our showroom.
Exhibition: MedtecLIVE from 30 June to 2 July 2020
Due to the current COVID-19 situation, MedtecLIVE, which was planned to take place in Nuremberg from 30 June to 2 July 2020, cannot be held in person. Medtec will now take place virtually via a platform.
You can register for participation under the link.
Further information can be found on the website.
As we already notified you in an earlier message, Früh Verpackungstechnik AG is doing its utmost to maintain our
production supply chain and minimize the impact of the coronavirus on our workforce.
New brand identity
The aim of our new future-oriented brand identity is to more clearly express our service capacity.
We have been finding the best solutions for the toughest packaging challenges since 1980. We ensure continuous improvement in all areas in order to help our customers move forward with an outstanding solution. This service capacity and our forward-looking perspective is expressed more clearly in our updated brand identity. In addition to a design refresh, we have adapted our slogan, which is now “protect to perform.” Behind this slogan is our belief that protection can always be increased. We are constantly improving our solutions for customers. To ensure that every packaged product can unfold its potential – and people can benefit from them without concern.
Exhibition: MedtecLIVE has been postponed
Because of the unusual situation caused by the spread of the coronavirus MedtecLIVE has been postponed. MedtecLIVE will now take place from 30 June to 2 July 2020 in Nuremberg. We are looking forward to welcome you at our booth 611 in hall 9.
You find more information here: https://www.medteclive.com/en
Due to current circumstances and the spread of the coronavirus the situation in Switzerland has been categorized as special in accordance with the Epidemics Act.
Früh Verpackungstechnik AG has assembled an internal team comprised of members of executive and senior management. This team meets at regular defined intervals, uses the latest information (TV, radio, newspapers, government authorities) to analyze the latest developments and their impact on our company in order to be able to respond quickly to changing circumstances and recommendations by the national and cantonal authorities.
All employees are being made aware of the situation with the aim of providing them with in-depth information about current events, the spread of the disease in neighboring countries and among their friends and family as well as correct hygiene measures both within the company and outside of it. The hygiene measures and travel guidelines issued by the Swiss Federal Office for Public Health must be observed.
At present, we are not seeing any negative consequences in the supply chain and in our production. As a precautionary measure, we are increasing our stocks of face masks and disinfectants. Additional hand disinfectant stations have been set up for employees and visitors. We are reducing visits to foreign countries as well as customer and supplier visitors to our offices to an absolute minimum, and we are also postponing meetings that have already been until a later date set if such meetings are not absolutely necessary.
The aim of these measures as well as the ongoing involvement of our employees is to prevent the spread of the virus internally. The regular meetings will enable us to adapt the measures quickly to current circumstances and are intended to ensure the continued maintenance of production at our company.
Exhibition: MedtecLIVE
The annual Medtec will take place in Nürnberg, Germany on March 31 until April 02, 2020. Früh Verpackungstechnik will be there with a new stand and its experts.
Exhibition: Pharmapack in Paris
The annual Pharmapack will take place in Paris on February 05 and 06, 2020. Früh Verpackungstechnik will be there with a new stand and its experts.
PFS Conference in London
Visit us at Europe’s leading PFS conference in London.
The annual PFS Conference will take place in London on January 15 and 16, 2020. Früh Verpackungstechnik will be there with a new stand and its experts.






















